PTPs are signaling molecules that regulate a variety of cellular processes including cell growth, differentiation, mitotic cycle, and oncogenic transformation. The protein encoded by this gene is a member of the protein tyrosine phosphatase (PTP) family. Protein tyrosine phosphatase, non-receptor type 1 3 (APO-1/CD95 (Fas)-associated phosphatase) The studies also suggest that this subunit may mediate the gene induction and cataleptic behavior induced by haloperidol. Knockout studies in mice suggest that this subunit may play an important role in regulating energy balance and adiposity. This subunit has been shown to interact with and suppress the transcriptional activity of the cAMP responsive element binding protein 1 (CREB1) in activated T cells. This subunit can be phosphorylated by the activated catalytic subunit. The protein encoded by this gene is one of the regulatory subunits. Four different regulatory subunits and three catalytic subunits have been identified in humans.
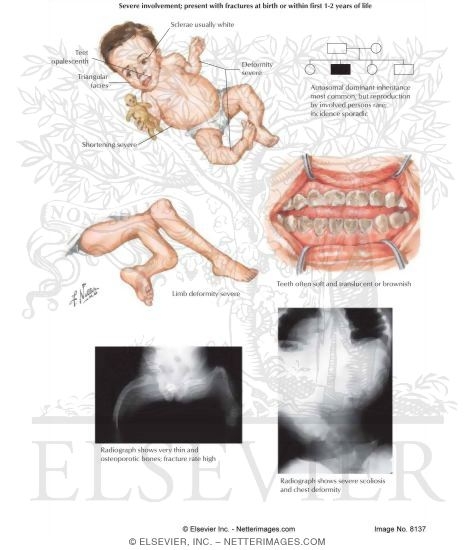
OSTEOGENESIS IMPERFECTA TYPE 3 FREE
cAMP causes the dissociation of the inactive holoenzyme into a dimer of regulatory subunits bound to four cAMP and two free monomeric catalytic subunits. The inactive kinase holoenzyme is a tetramer composed of two regulatory and two catalytic subunits. cAMP exerts its effects by activating the cAMP-dependent protein kinase, which transduces the signal through phosphorylation of different target proteins. Protein kinase, cAMP-dependent, regulatory, type II, betaĬAMP is a signaling molecule important for a variety of cellular functions. This subunit has been shown to regulate protein transport from endosomes to the Golgi apparatus and further to the endoplasmic reticulum (ER). It may interact with various A-kinase anchoring proteins and determine the subcellular localization of cAMP-dependent protein kinase. Protein kinase, cAMP-dependent, regulatory, type II, alphaĬAMP is a signaling molecule important for a variety of cellular functions.
